e_NanoParticles
Plasma-induced discharge electrolysis to form nanoparticles
Even when one electrode is outside the electrolytic bath, a plasma-induced stationary discharge allows electrolysis under certain conditions. The principle of the process is shown in Fig. 7. A photo inside the electrolytic cell under stationary discharge is also shown in Fig. 7.
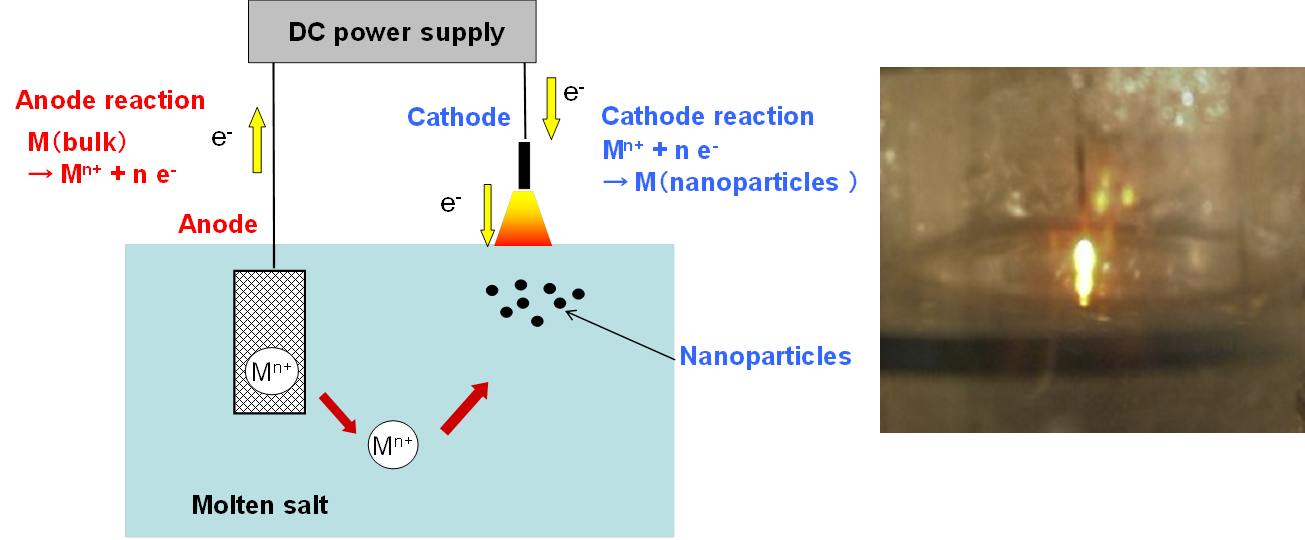
Fig. 7 Principle of plasma-induced discharge electrolysis
With this discharge electrolysis method so far, we have obtained many types of metal and alloy nanoparticles, including Si, Ti, Fe, Ni, Co, Zr, Nb, Ta, W, Ag, and Pt as well as various alloys. To continuously obtain finer and more uniform nanoparticles, electrolysis with a rotating disk is available. The cell’s scheme is shown in Fig. 8.
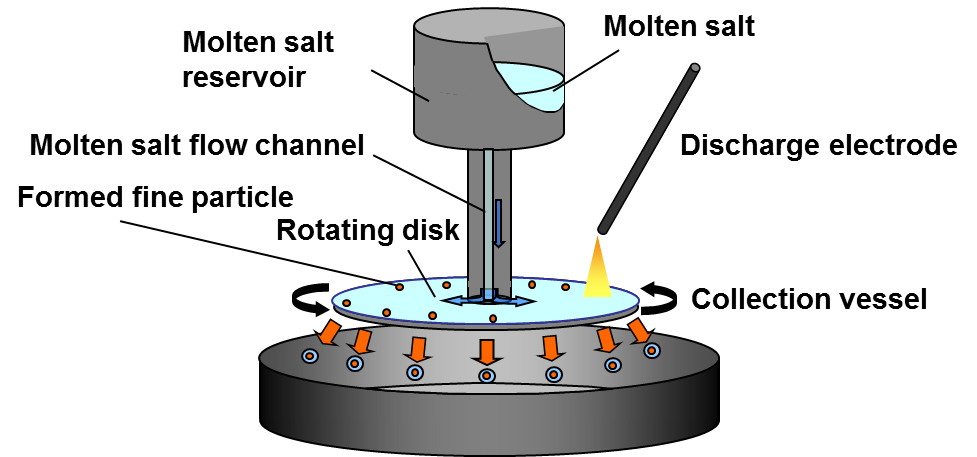
Fig. 8 Scheme of cathodic discharge electrolysis using rotating disk
With such a cell, where a rotating Ni disk anode was adopted, we successfully obtained Ni nanoparticles whose diameter is less than 10 nm at 2,500 rpm (Fig. 9).
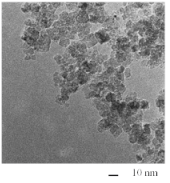
Fig. 9 TEM Images of Ni nanoparticles formed by plasma-induced cathodic discharge electrolysis using rotating disk anode-type electrolytic cells
We successfully carried out scale-up experiments by selecting Ta nanoparticle production as an example (production capacity: 200g/day). The production of finer and uniform Ta nanoparticles is needed for a high power and high energy density tantalum capacitor that is anticipated for future power electronics devices. With this process, recycling and effectively reusing Ta can be attained simultaneously.
Note that this process is also available for important metals other than Ta, including precious and refractory metals.
