e_CarbonFilm
Electrochemical formation of carbon film and its applications
The electrochemical formation of carbon film can be achieved either by the anodic oxidation of C22- ions or by the cathodic reduction of CO32- ions. The principle of the electrochemical formation of carbon film by the anodic oxidation of C22-ions is shown in Fig. 2.
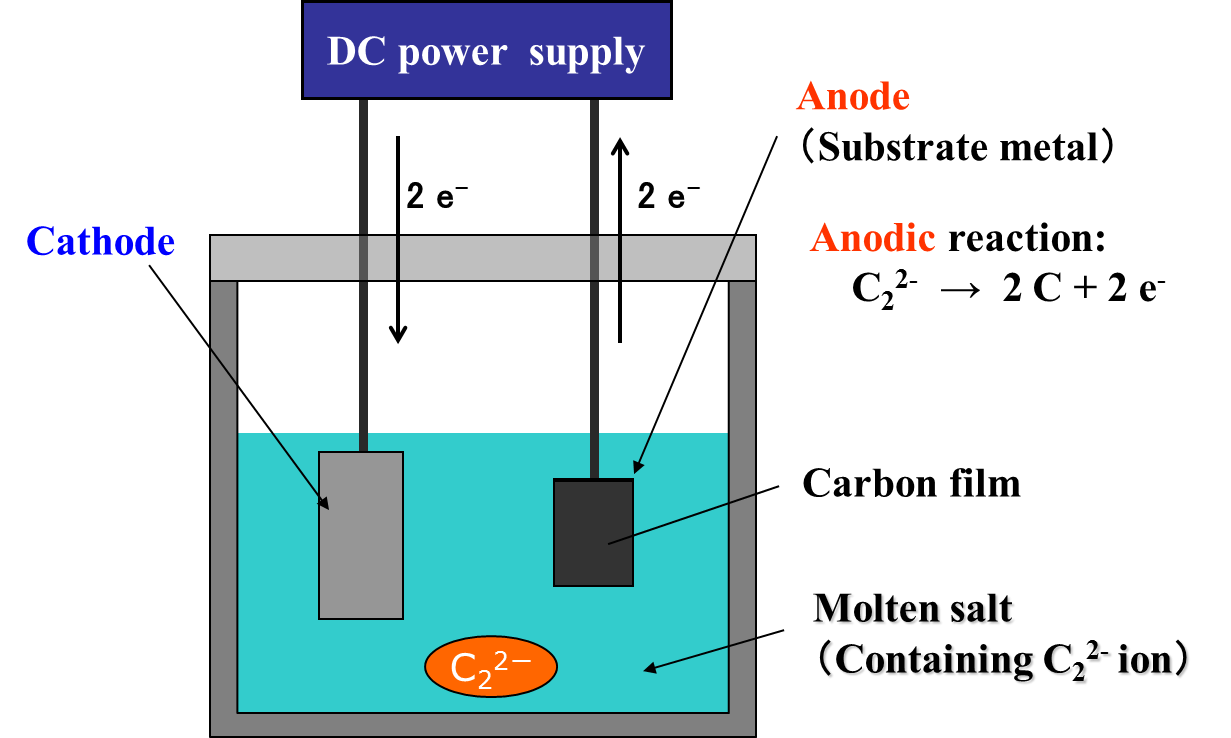
Fig. 2 Principle of carbon film formation by anodic oxidation of C22- ions
Coating with dense and adherent carbon film can be achieved by the anodic oxidation of C22- ions (Fig. 3).

Fig. 3 Dense and adherent carbon film electrochemically formed on stainless steel
They show high corrosion resistance against various types of corrosion tests. Since the contact electrical resistance measurement of a carbon-coated metal plate also showed very promising results, the obtained carbon-coated metal actually has advantageous features for a fuel cell separator, a current collector of a Li-ion secondary battery/capacitor, and as a substitute for the fluoric resin film used in semi-conductor/liquid crystal production lines.
Since we can even coat the surface of a screw thread by a thin and dense adherent carbon film as well as the inside of a porous body (Fig. 4), this process is also available for producing corrosion-resistant fastening components (bolts, nuts, and many other applications).

Fig. 4 Carbon film on various substrates: (a) screw threads and (b) porous nickel
On the other hand, carbon film can be obtained by a cathodic reduction of a CO32- ion. The principle of carbon film formation by a cathodic reduction of CO32- ions is shown in Fig. 5.
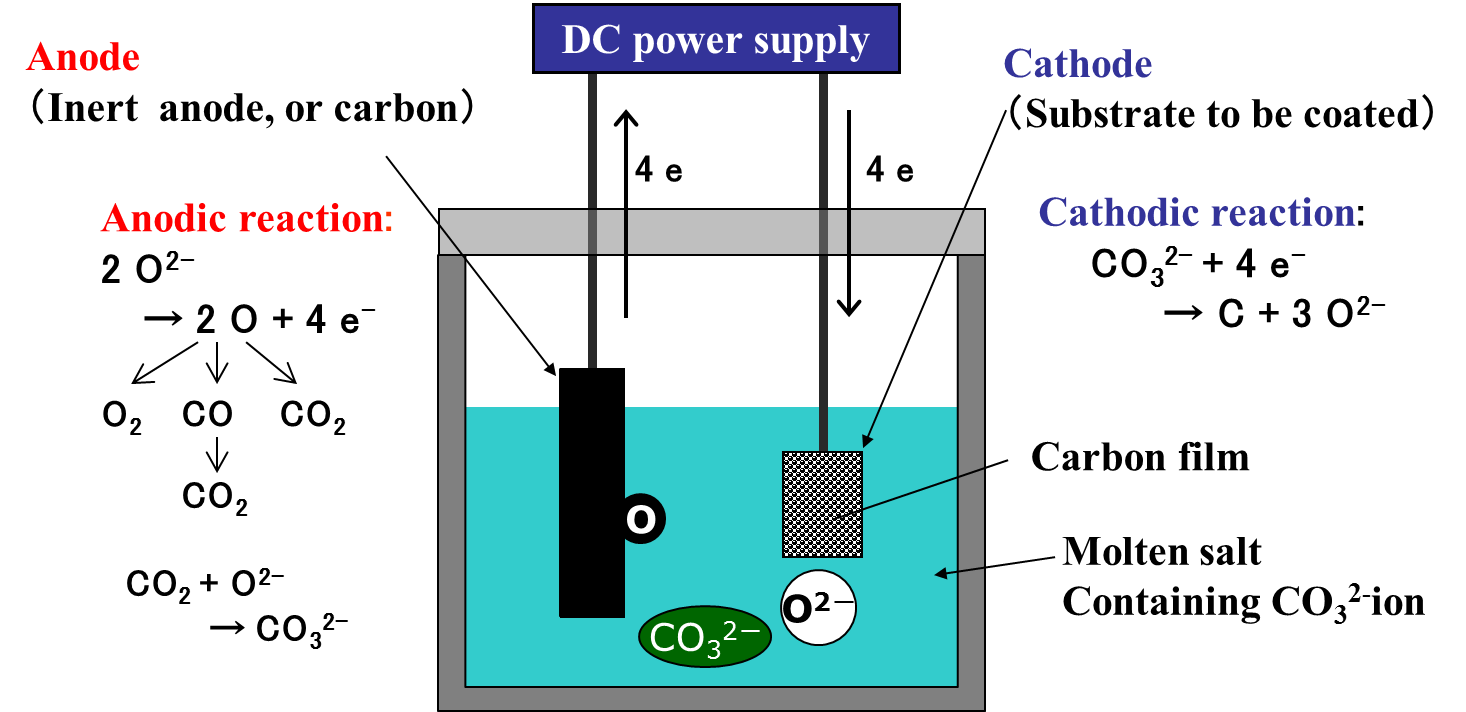
Fig. 5 Principle of carbon film formation by cathodic reduction of CO32- ions
The carbon film obtained by a cathodic reduction of CO32- ions has a micro-porous structure (Fig. 6), which is advantageous for applications to an electric double-layer capacitor or a super-capacitor.

Fig. 6 Carbon film electrochemically formed on Al
It is also possible to electrochemically form a porous carbon film on a very thin, dense, and adherent carbon film that is electrochemically formed on the metal surface beforehand. The formation of such a thin, dense, and adherent surface coating film can be achieved, for instance, by applying either a potentiostatic pulse or a periodically reverse (PR) electrolysis.
